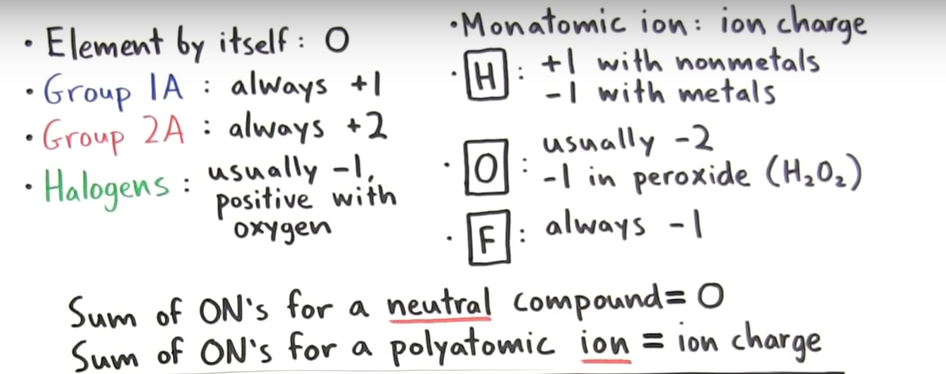Chemistry 12
Absences: It is expected that in the case of absence, the student is responsible for catching up missed material online. The course website contains ALL worksheets, notes, videos and answer keys needed to make up absences from offsite. If a student misses a quiz, test or lab due to an excused absence validated by a parent or guardian, it is the student’s responsibility to re-schedule the missed assessment as soon as possible outside regular class hours.
|
Course Outline LINK
Assessment
|
TIMELINE
Unit 0: Introduction, Review, Safety 1wk Unit 1: Reaction Kinetics 3wks Unit 2: Equilibrium 3wks Unit 3: Solutions and Equilibrium (spring break) Midterm Exam Unit 4: Acids, Bases 3wks Unit 5: Oxidation-Reduction 3wks Unit Review: 2wks Final Exam |
Chemistry 11 review
Chemistry 11 Review | ***up to +5% midterm grade***| Due February 2 2024
CHEM 11 REVIEW VIDEO
Chem. 11 review part 1 part 2 part 3 part 4 part 5 part 6
RECALL:
Endothermic vs Endothermic Rxns. VIDEO
How to predict products VIDEO
Types of Chemical Reactions. IMAGE
Basic Stoichiometry VIDEO
Limiting & Excess reactants. VIDEO
Molarity Stoichiometry. VIDEO
Solution Stoichiometry. VIDEO
VIDEO 1: naming ionic and molecular compounds
VIDEO 2: naming compounds with polyatomic ions
VIDEO 3: naming acids
TOPICS: The Scientific Method, Safety, Science 10 review, Unit A House keeping Powerpoint LINK Sciences Song link The Scientific Method LINK VIDEO VIDEO. Activity LINK. NOTES
Lab Safety VIDEO link Procedures link Safety Contract link Safety Map link Safety song link
CHEM 11 REVIEW VIDEO
Chem. 11 review part 1 part 2 part 3 part 4 part 5 part 6
RECALL:
Endothermic vs Endothermic Rxns. VIDEO
How to predict products VIDEO
Types of Chemical Reactions. IMAGE
Basic Stoichiometry VIDEO
Limiting & Excess reactants. VIDEO
Molarity Stoichiometry. VIDEO
Solution Stoichiometry. VIDEO
VIDEO 1: naming ionic and molecular compounds
VIDEO 2: naming compounds with polyatomic ions
VIDEO 3: naming acids
TOPICS: The Scientific Method, Safety, Science 10 review, Unit A House keeping Powerpoint LINK Sciences Song link The Scientific Method LINK VIDEO VIDEO. Activity LINK. NOTES
Lab Safety VIDEO link Procedures link Safety Contract link Safety Map link Safety song link
INTRODUCTION & SAFETY
|
POWERPOINT LINK
Sciences Song link Safety Science Equipment WS Resources The Scientific Method Units Conversion Sheet Data Booklet Resource Mole Bridge pH/pOH Hydrolysis Chemistry Glossary |
|
|
QUIZ 1 Friday January 26
UNIT 1: REACTION KINETICS Hebner p. 1-36
Reactants must collide to react, and the reaction rate is dependent on the surrounding conditions.
|
Feb 8Hebden p. 1-10 # 1-17
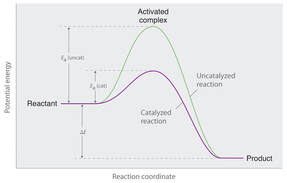
NOTES: Reaction Rates VIDEO 1 2 WS: Rates of Reaction | KEY VIDEO NOTES: Factors Affecting Reaction Rates WS: Reaction Rates | KEY IMAGE 1 2 NOTES: Collision Theory VIDEO QUIZ 2 Feb 2 NOTES: Energy Changes in Chemical Reaction LAB: Reaction Kinetics Activity Sheet NOTES: Activation Energy VIDEO Hebden p. 20-25 # 35, 36, 41-45 NOTES: Reaction Mechanisms & Catalysts. VIDEO * WS: Reaction Mechanisms | KEY Hebden p. 26-35 # 54-57, 61 Quiz 2 Thursday Feb 8 Unit Review LINK l KEYFeb 9, 13 Lab report l KEY. l PART D l Feb 6 + 9, due Feb 29 Title page (Title, date, name, names of partners, picture?) 4 Introductions (balanced chemical equation, mechanisms where applicable, basic info on endo/exo, phases, safety, variables), 4 hypothesis, Purposes, Materials, Procedures, Data Recordings, Analysis, Discussions (sources of error, observations of colour, gas, heat, safety, 4 conclusions Formal Lab report Rubric UNIT TEST Feb 15 RETEST Friday Feb 19 at lunch room 141 |
|
|
UNIT 2: EQUILIBRIUM Hebden p. 37-72
Dynamic equilibrium can be shifted by changes to the surrounding conditions.
|
NOTES: Reversible Reactions
NOTES: Predicting Spontaneous Reactions. VIDEO DO p. 48 #15 WS1 -Equilibrium, Enthalpy, Entropy | KEY NOTES: Le Châtelier's Principle VIDEO l VID part 1 l part 2 WS2-Le Châtelier | KEY NOTES: Equilibrium Expression NOTES: Equilibrium Calculations WORKSHEETS: Graphically Represented WS | KEY Spontaneous, Non-Spont, or Equilib. WS | KEY Le Châtelier Shifts WS | KEY Hebden p. 64-65 # 17-30 ( extending # 31-35 p. 60) Quiz Friday Mar 1 Eq Expression & Eq Constant WS | KEY ICE tables VIDEO at 39:19sec. and PAGE Eq Calculations WS | KEY UNIT REVIEW: ws l KEY or WS | KEY TEST: Tuesday March 5th HEBDEN:
p. 37-39. Intro to Dynamic Equilibrium. VIDEO p. 40-43 The Characteristics of Equilibrium VIDEO p. 43-49. Predicting Spontaneous Reactions. VIDEO VIDEO p. 50-55. Le Chatelier's Principal. VIDEO p. 57-60. Eq. Expression and Constant. VIDEO. VIDEO p. 61-71 Le Chatelier's Principal and Eq. Constant. VIDEO |
|
UNIT 3: SOLUBILITY EQUILIBRIUM Hebden p. 73-105
Saturated solutions are systems in equilibrium.
Due
|
TAKE-HOME UNIT DUE MARCH 25 10% FINAL GRADE
NOTES: Intro to Solubility p. 73-76 Intro to Solubility WS l KEY. or #1-7. NOTES: Calculating Solubility & Ion Concentration. p. 77-81 Solubility Concepts WSl KEY or #8-16 +18-20. NOTES: Predicting Solubility. p. 81-84 Predicting Solubility WS l KEY or #21-24 NOTES: Formula, Complete & Net Ionic Equations p. 84-88 Writing Equations WS l KEY or # 25 NOTES: Qualitative Analysis. p. 88-91 Qualitative Analysis WS l KEY or #26-36 LAB: Qualitative Analysis | Rubric NOTES: Solubility Product. p. 91-95 Qual. Analysis & Ksp Calculations WS l KEY or #41-55. NOTES: Predicting Whether a Precipitate will Form. p. 96-99 Do Hebden # 56-69 NOTES: Le Châtelier & Solubility p. 99-102 Le Châtelier & Solubility WS l KEY. or #70-75. Removing Pollution/ Hardness p. 102-104 # 76-80 Common Ion Effect p. 105-108 #81-86 LAB: Determination of a Solubility Product Constant Take-home assignment due March 31 worth 10% of grade Unit 3 Take Home 1: Solutions Equilibrium l Key Unit 3 Take-Home 2: Solutions Equilibrium l Key |
|
Midterm Units 1-3 April 4
Unit 1 Reaction Kinetics Review and Key LINK
Unit 2 Equilibrium Review and Key LINK
Unit 3 Solutions Review and Key LINK
Unit 2 Equilibrium Review and Key LINK
Unit 3 Solutions Review and Key LINK
UNIT 4: ACIDS & BASES Hebden p. 109-184 March 25-April 25
Acid or base strength depends on the degree of ion dissociation.
|
Intro video 1 video 2. CRASH COURSE VIDEO
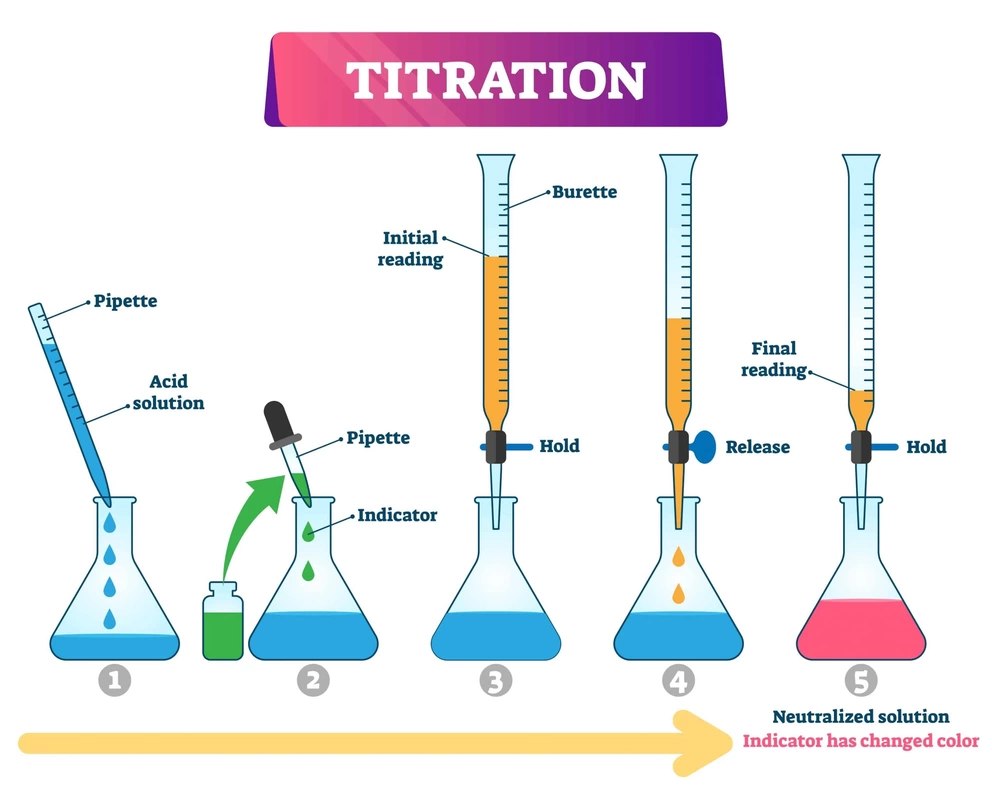
Recall Acid Naming LINK & common acids LINK REVIEW Unit 3 net ionic equations p. 84 REVIEW Titration VIDEO VIDEO p. 112 NOTES1: Characteristics of Acids & Bases Common Acids & Bases WS | KEY p. 116 NOTES2: Brønsted-Lowry Theory l VIDEO l VIDEO Brønsted-Lowry Acids & Bases WS | KEY p. 121 NOTES3: Strengths of Acids & Bases l VIDEO Strengths of Acids & Bases WS | KEY p. 126 NOTES4: Kw l VIDEO Kw Calculations WS | KEY Review for Quiz: VIDEO Net ionic equations VIDEO Hebden p. 109-123, Worksheets, Net ionic equations VIDEO QUIZ 1 Friday April 5 Acids/Bases, Strengths , Conjugates p. 134 NOTES5: pH and pOH l Intro VIDEO l VIDEO l FLOW CHART p. 137-139 # 49-53 pH and pOH Calculations WS l KEY p. 141 NOTES6: Mixing Strong Acids & Bases l VIDEO l VIDEO Mixing Strong A & B | KEY more examples LINK #5,7,8,11,13 p. 128 NOTES7: Ka/b Acid & Base Equilibrium Constants l VIDEO Acid & Base Constants Ka/b WS | KEY EXTRA PRACTICE & KEY WS REVIEW WS l KEY Quiz 2: Kw & Ka/Kb & pH/pOH THU p. 144. NOTES8: Hydrolysis lVIDEO 1 and 2 Flow Chart VIDEO Hydrolysis WS | KEY Don't do 6, 9, 10. p. 148. NOTES9: Ka & Kb Calculations l VIDEO @4:32 FULL VIDEO Ka & Kb Calculations WS | KEY CHOOSE ANY 5 p. 154. NOTES10: Acid-Base Titrations VIDEO VIDEO Acid-Base Titration WS | KEY. NOT # 7 or 9 p. 159. NOTES11: Indicators VIDEO Indicators WS | KEY Don't do: 1 d, e, 5b, 6b, e, 7c. p. 164-177 NOTES12: Titration Curves do # 121-130 LAB: Titration IMAGE lab LINK RUBRIC DUE May 15 RESUBMIT BEFORE MAY 31 % error calculation: (experimental value-given value)/given value X 100p. 186. Acid Rain WS | KEY VIDEO LINK Buffers LINK p. 177. NOTES13: Buffers QUIZ # 3 l KEY Review Unit 4 l KEY PowerPoint MORE Review with key LINK (don't do #22, 32) Practice test and solutions TEST l key RETEST EXTRA Review 1 and 2l KEY 1 and 2 LAB: Hydrolysis optional HYDROLYSIS EXTRA WS & key l Classification & key REVIEW NOTES Hebden:#: 67, 69.e), 99, 101, 104, 113, 115, 118, 120, 125.e), 126, 130, 139, 140 |
|
Unit 5: OXIDATION-REDUCTION. Hebden p. 189-246
Oxidation and reduction are complementary processes that involve the gain or loss of electrons.
|
What is a redox reaction? LINK
Electrochemistry Summary NOTES1: Intro to Electrochemistry & Oxidation Numbers Intro to Redox WS | KEY Intro VIDEO l Oxid. # VIDEO VIDEO WS 1 #1-8 May 16 p. 192-195 # 1-5 in Hebden NOTES2: Predicting Spontaneous Reactions. VID. VID Predicting Spontaneous Reactions WS | KEY p. 195-200 in Hebden # 7-18 EVENS ONLY. VIDEO Quiz 1 Redox, oxidation #, Spnt/ non spont rxn NOTES3: Balancing Half Reactions VIDEO Balancing Half Reactions WS | KEY QUIZ 2 Balancing half reactions p. 205 in Hebden NOTES4: Balancing Redox Equations. VIDEO Balancing Redox Equations WS | KEY REVIEW l KEY DO #1-14 Unit 5 test _________________________________________ LAB: Redox Titration | Rubric NOTES5: Electrochemical Cells Electrochemical Cells WS | KEY NOTES6: Standard Reduction Potentials Standard Reduction Potentials WS | KEY NOTES7: Selecting Preferred Reactions NOTES8: Electrolysis Electrolysis WS | KEY NOTES9: Corrosion of Metals Unit Review: |
|
FINAL EXAM January 2024
|
FINAL REVIEW
FINAL REVIEW NOTES LINK FINAL EXAM Review and key OLD Exam MANY PRACTICE FINALS & KEY LINK MORE...Chem 12 study notes. HERE Chemistry 12 Final Study Guide 1994 Final exam Chem 12 Calculations POwerPOint Chem 12 Review video playlist link |
OLD BC EXAMS BRAIN GENIE APP
|